Microplastics are ubiquitous. They result from the breakdown of larger plastic waste (plastic bottles, bags, straws, glitter, fishing nets, toothbrushes, etc.) as well as synthetic fabrics (spandex, nylon, polyester, etc.). Research has discovered these micropollutants in our oceans, our shellfish, and our bottled water. However, these tiny plastic particles can be challenging to study and therefore remain vastly uncharacterized.
Methodologies for the detection, characterization, and quantification of microplastics continue to be developed. In response to the need to automate microplastic characterization, some researchers have turned to FlowCam imaging flow cytometer to image, characterize, and quantify them. The FlowCam was used in two recent studies to quantify, image, and measure microplastics.
Microplastics in Blue Mussels
Woods et al. (2017) from the Marine and Environmental Research Institute (Blue Hill, Maine) and Bigelow Laboratory for Ocean Sciences (Boothbay, Maine) quantified the uptake, digestion, and egestion of polyethylene terephthalate microplastic fibers (MPF) in blue mussels (Mytilus edulis) using the FlowCam.

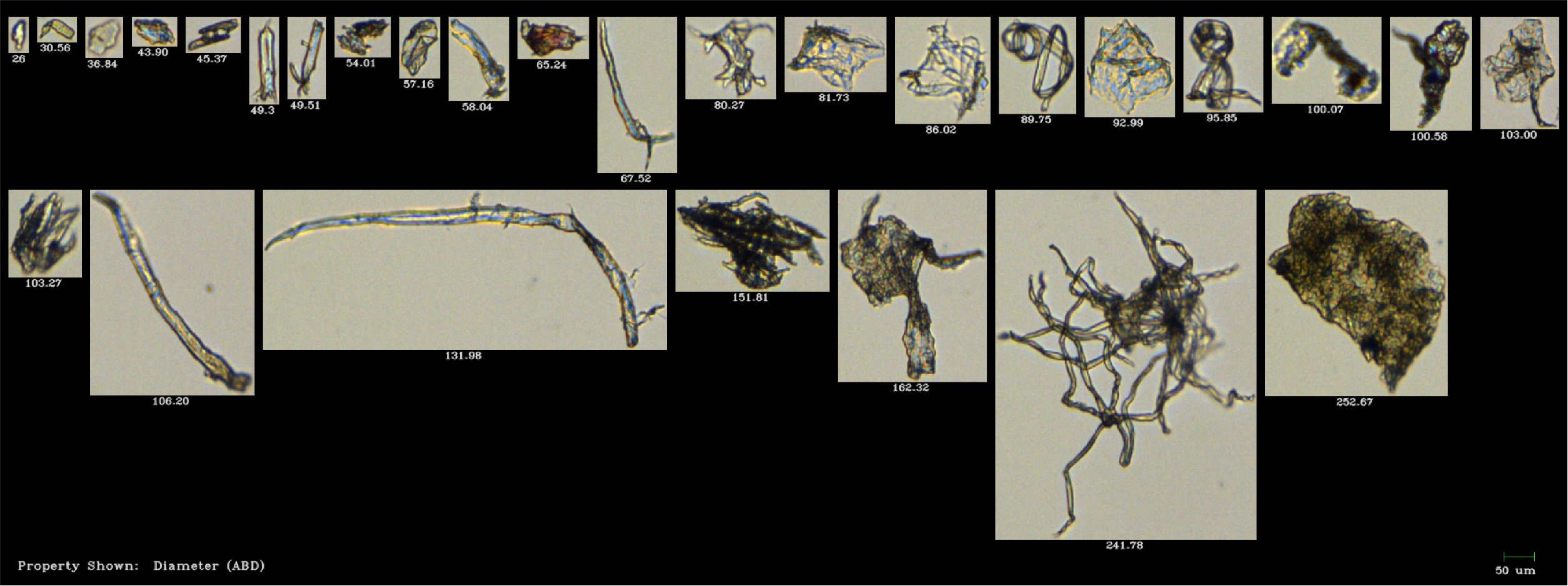
Pictured above: Polyethylene terephthalate microplastic fibers (MPF) as imaged by the FlowCam. The crisp, non-weathered edges of the above MPF distinguished these from the naturally occurring, weathered fibers from the lab environment during the study. Credit: Woods et al. (2017).
Woods et al. found that microalgal uptake rates were greatly reduced in mussels exposed to concentrations of 15,000 MPF/L or greater. Pseudofeces production showed a positive correlation with MPF uptake rates at 30,000 MPF/L. A single fecal pellet showed up to 70 MPF.
Better Microplastic Research Using Digestion
Lorenz et al. (2017) from the Alfred Wegener Institute for Polar and Marine Research evaluated the efficacy of microplastic isolation in a natural marine water sample using a six-step enzymatic oxidative digestion developed by Löder et al. (2017).
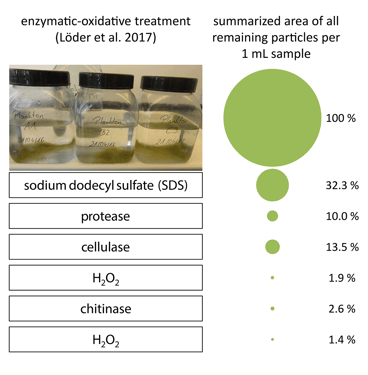
Lorenz et al. observed that the complete digestion process (all six steps) resulted in a 98.6% reduction in total particle area. Following the digestion, the remaining particulate matter was filtered from the water matrix. The total area of particulate matter per mL of sample was calculated using the FlowCam to determine an appropriate volume to be filtered so as not to overburden the filter.
Above: The six-step digestion used by Lorenz et al. to isolate microplastics from organic matter in marine water samples. Each step shows a reduction in the area of remaining particulate matter, as calculated by VisualSpreadsheet using FlowCam images. Credit: Lorenz et al. (2017).
Read about the studies of Woods et al. and Lorenz et al. in detail. Download the poster presented at the 2018 Ocean Sciences Meeting in San Diego, California.
Learn more about how FlowCam is used for aquatic research around the globe, from microplastic characterization to phytoplankton population monitoring.
References:
Woods, M.N., Stack, M., Fields, D.M., Matrai, P., (2017) Microplastic fber uptake, ingestion, and egestion rates in the blue mussel (Mytilus edulis). Unpublished raw data, Marine and Environmental Research Institute, Blue Hill, ME, and Bigelow Laboratory for Ocean Sciences, Boothbay, ME.
Lorenz, C., Speidel, L., Primpke, S., Gerdts, G., (2017) Using the FlowCam to Validate an Enzymatic Digestion Protocol Applied to Assess the Occurrence of Microplastics in the Southern North Sea. MICRO 2016: Fate and Impact of Microplastics in Marine Ecosystems doi: 10.1016/B978-0-12-812271-6.00094-6.
Löder, M.G.J., Imhof, H.K., Ladehoff, M., Löschel, L.A., Lorenz, C., Mintenig, S., et al. (2017) Enzymatic Purifcation of Microplastics in Environmental Samples. Environ Sci Technol 51:14283-14292 doi:10.1021/acs.est.7b03055.